Ophthalmic
Low-Volume Sterile Preparations for Ocular Use.
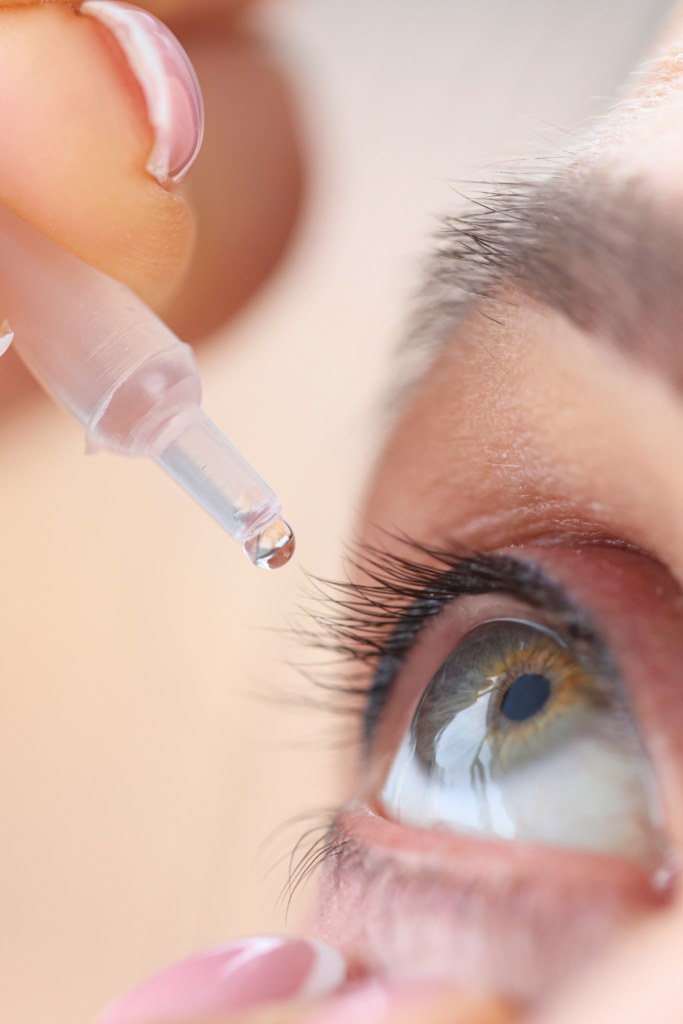
Compounded solutions and suspensions produced under aseptic conditions with container-closure considerations for ocular delivery.
What We Compound (examples)

1
Dosage Forms & Packaging
Low-volume sterile droppers; concentration, vehicle, and preservatives tailored to patient and prescriber requirements.
2
Quality & Documentation
Sterile compounding in ISO-classified cleanrooms; environmental monitoring, filter integrity controls, and documented workflows. Third-party sterility/endotoxin/potency testing available; BUD and labeling guidance where applicable.
Get in Toutch
Compounded medications are prepared for individual patients pursuant to a valid prescription. They may not be reviewed or approved by the FDA. Availability varies by state licensing. Testing availability and turnaround can vary by product.
